- Blog
- Metro exodus skidrow
- Official arduino usb host shield
- Watch the curse of la llorona
- The constant gardener movie online dailymotion
- How many speeds does acid music studio 10 has for burning
- Ivipid fox logo
- Atlas lathe change gears
- Japji sahib audio punjabi
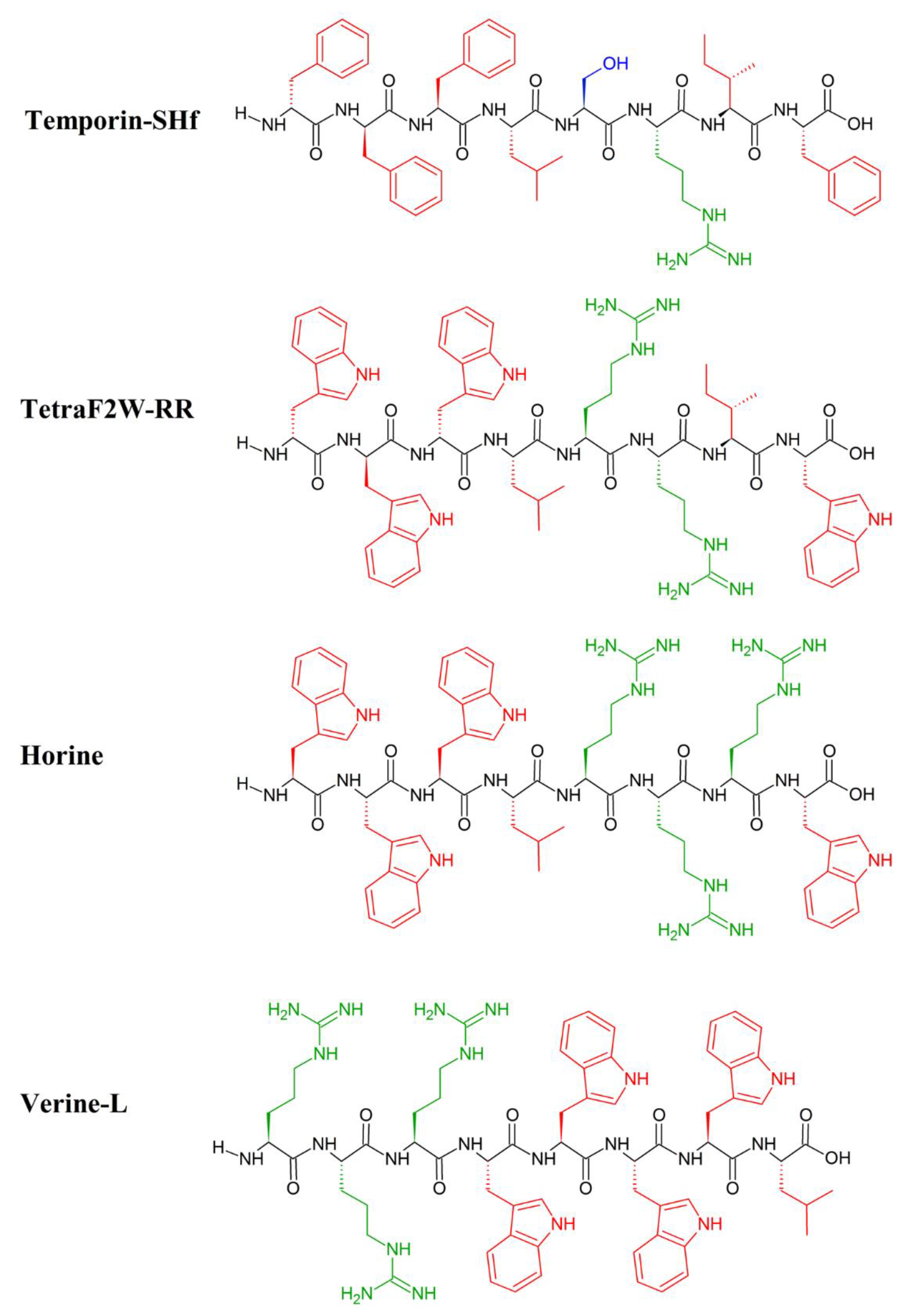
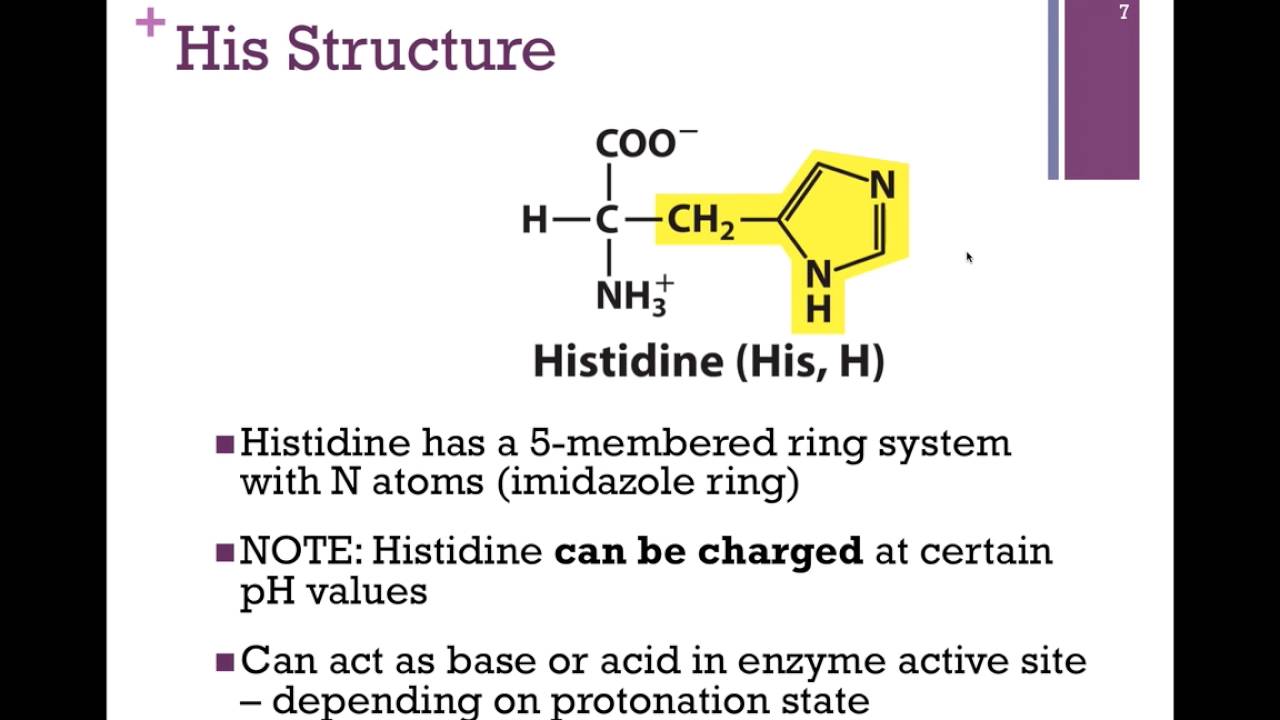
- Hydrophilic vs hydrophobic amino acids
- Netflix account
- Kernel mode driver framework 1-11 download windows 7 64 bit
- Puffin browser for pc windows 7 free download
- Orcaflex web review
- Download usb advance ps2
It gets more complicated when things are moving - as is often the case in real-world situations. (There are exceptions, however, where special kinds of patterning can actually reverse a material’s ordinary properties.) Similarly, nanopatterning of a hydrophilic surface can make it superhydrophilic. The shape of a surface can also amplify the effects: For example, if a material is hydrophobic, creating nanopatterns on its surface can increase the contact area with a droplet, amplifying the effect and making the surface superhydrophobic.
Why do these phenomena happen? It’s essentially a matter of surface chemistry, which is determined by the characteristics of the materials used. Conversely, for applications where water flows over a surface to keep it from overheating, it’s desirable to have a superhydrophilic material, to assure maximum contact between the water and the surface. For example, the surfaces of condensers in desalination plants or power plants work best when they are superhydrophobic, so droplets constantly slide off and can be replaced by new ones. “In a lot of cases, it’s the extreme behavior that’s useful in engineering,” says Evelyn Wang, an associate professor of mechanical engineering at MIT who specializes in superhydrophobic materials. If the droplets are spread out nearly flat, with a contact angle of less than about 20 degrees, the surface is superhydrophilic. Though the definitions of these terms are less precise, surfaces where tight droplets form a contact angle of more than 160 degrees are considered superhydrophobic. But if the droplet forms a sphere that barely touches the surface - like drops of water on a hot griddle - the contact angle is more than 90 degrees, and the surface is hydrophobic, or water-fearing.īut the terminology doesn’t stop there: Most current research on hydrophobic and hydrophilic materials is focused on extreme cases - namely, superhydrophobic and superhydrophilic materials.
If the droplet spreads, wetting a large area of the surface, then the contact angle is less than 90 degrees and that surface is considered hydrophilic, or water-loving (from the Greek words for water, hydro, and love, philos). Hydrophilic and hydrophobic materials are defined by the geometry of water on a flat surface - specifically, the angle between a droplet’s edge and the surface underneath it. Photo courtesy of Rong Xiao and Nenad Miljkovic
- Blog
- Metro exodus skidrow
- Official arduino usb host shield
- Watch the curse of la llorona
- The constant gardener movie online dailymotion
- How many speeds does acid music studio 10 has for burning
- Ivipid fox logo
- Atlas lathe change gears
- Japji sahib audio punjabi
- Hydrophilic vs hydrophobic amino acids
- Netflix account
- Kernel mode driver framework 1-11 download windows 7 64 bit
- Puffin browser for pc windows 7 free download
- Orcaflex web review
- Download usb advance ps2
